Asghar Razavi, Ph.D. 1300 York Ave, New York, NY, 10044
Asghar Razavi
This website illustrates my academic research during my postdoctoral work with Harel Weinstein at Weill Cornell Medical College and PhD work with Vincent Voelz and Temple university.
My postdoctoral academic research was focused on understanding function of membrane proteins that clear neurotransmitters from synaptic cleft. These family of proteins are called neurotransmitter:sodium symporters (NSS) since their function depends on electrochemical gradient of sodium ions across membrane. Important members of NSS family include dopamine transporter (DAT), serotonin transporter (SERT), and norepinephrine transporter (NTE) that remove dopamine, serotonin, and norepinephrine from synaptic cleft. Proper control of the amount of neurotransmitter in the synapse is important for many brain functions and imbalances are linked to brain related diseases like depression, bipolar disorders, ADHD, Schizophrenia, and Parkinson’s. In my research, I combine MD simulations with kinetic models based on Markov State Models (MSMs) to understand function of NSS family of proteins.
You can find more detailed information about my research endeavors by selecting specific topics under the “Current Research” or “Past Research” tabs in this website.
Curriculum Vitae (PDF)
Research
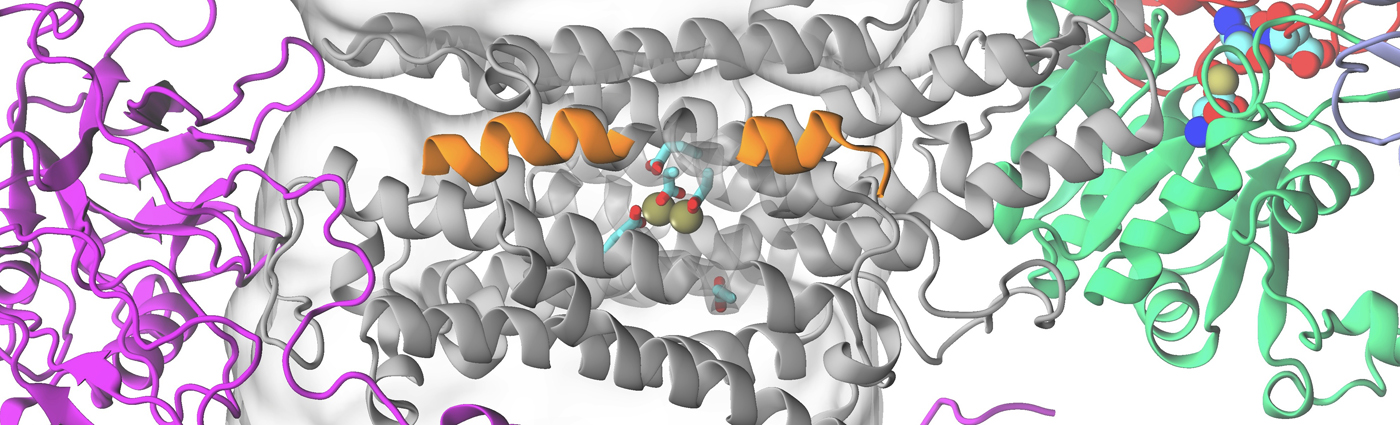
Na+/K+-ATPase
Na+/K+-ATPase (NKA) is a membrane protein that transports Na+ ions out of the cell and brings K+ ions into the cell against their concentration gradient. To function, NKA harnesses the chemical energy stored in an ATP molecule to cycle between two major conformational states during active pumping: a high affinity state for Na+, and a high affinity state for K+. The recent availability of the crystal structures [1] for these states, now makes it possible to computationally determine the factors that control selectivity for Na+ versus K+ ions. High selectivity of NKA for Na+ or K+ may be a result of thermodynamic (different affinities for binding sites) as well as kinetic (barriers along binding pathways) factors. To address the thermodynamic